The Thyroid Gaslighting
Why “normal” labs and a functioning thyroid are not the same thing
She sits across from me with a folder. Lab reports, printed in triplicate, flagged and annotated in the margins. Three years of TSH values, all landing squarely in the “normal” zone. She is gaining weight despite the kind of dietary discipline most people cannot sustain. She is losing hair. She sleeps nine hours and wakes exhausted. She struggles with a cognitive fog her colleagues have not noticed yet, but she has, acutely, because she used to be faster.
She has been offered antidepressants twice. She declined both times.
I notice she has never had a complete thyroid panel. Just TSH, Total T4, and Free T4. No antibody testing, and she was never told to stop all biotin for 48 hours prior to the blood tests (and biotin was in her daily multivitamin).
Biotin interferes with thyroid testing, potentially falsely lowering TSH and falsely elevating T4 and T3 levels.
What follows is what I wish every physician understood before they handed a woman like this a prescription for an SSRI and called it a day.
The TSH Monoculture
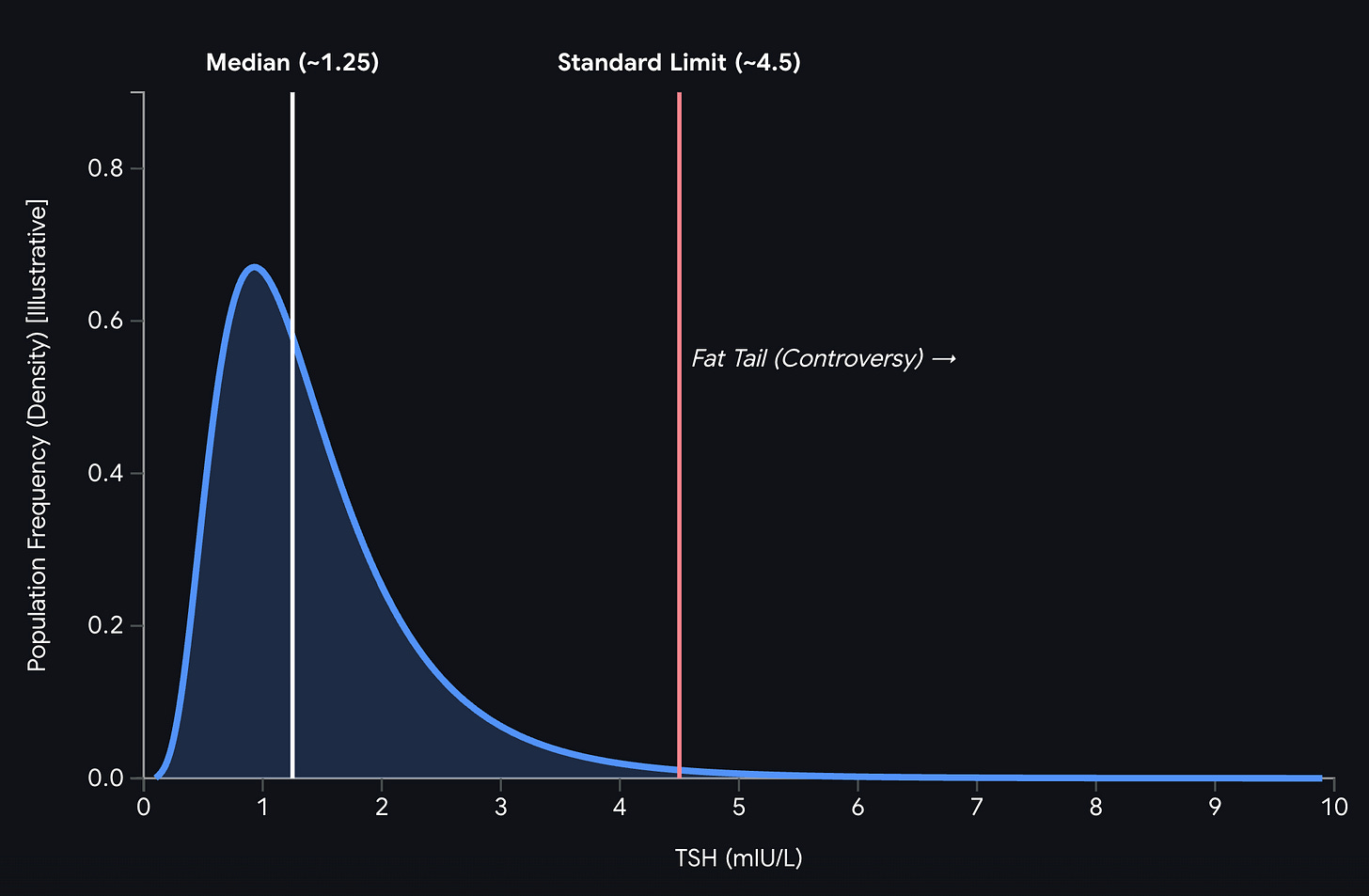
Modern medicine’s approach to thyroid health has calcified around a single number. Thyroid-stimulating hormone, or TSH, has become the near-exclusive arbiter of whether a thyroid is “fine.” If TSH falls within the reference range of 0.45 to 4.5 mIU/L, the diagnostic inquiry ends. The patient is reassured, the chart is closed, and the conversation about her symptoms pivots elsewhere, usually toward stress, sleep hygiene, or mood.
This is not medicine. It is population management.
The reference range itself is a statistical artifact, not a biological truth. It was derived primarily from NHANES III, a massive U.S. population dataset of over 17,000 individuals. The reference subset, those presumed disease-free, had a geometric mean TSH of 1.40 mIU/L. The actual 97.5th percentile of that rigorously screened subgroup was 4.12, not 4.5. The upper bound used by most commercial labs did not come from the study’s own data but from the assay manufacturer’s reference range. The number most physicians treat as biological gospel was set by the company selling the test.
It gets worse. Even within that “disease-free” reference population, the TSH distribution was not Gaussian. It was right-skewed, with a long upper tail. The reason is that antibody-negative Hashimoto’s thyroiditis is real. Thyroid ultrasound can reveal structural changes due to lymphocytic infiltration before antibodies are ever detectable in the blood. The reference population was contaminated from the start by the very disease it was supposed to exclude.
TSH tells you the thermostat is set. It does not tell you whether the heat is reaching the rooms.
The National Academy of Clinical Biochemistry eventually concluded, after rigorous exclusion criteria, that over 95% of truly healthy individuals maintain a TSH below 2.5 mIU/L. The American Association of Clinical Endocrinologists formally proposed narrowing the clinical reference range to 0.3 to 3.0. Neither recommendation was adopted by the mainstream bodies. The range stayed. The patients stayed sick.
A TSH of 2.8, the number my patient had been reassured by for three years, places her outside the optimal range of a rigorously healthy population. It is a signal, not a clearance.
And even if the number were perfect, it would still be measuring the wrong thing. TSH reflects the hormonal status of one tissue: the anterior pituitary. It does not measure free T3, the biologically active hormone. It does not measure peripheral conversion efficiency. It does not measure what is happening in the liver, brain, cardiac muscle, or skeletal tissue. The pituitary contains approximately 1,000 times more type-2 deiodinase activity than peripheral tissues and essentially no type-3 deiodinase activity, the enzyme responsible for converting T4 into the inactive reverse T3. The pituitary is uniquely privileged. It can remain satisfied while the rest of the body starves.
The Conversion Problem Nobody Talks About
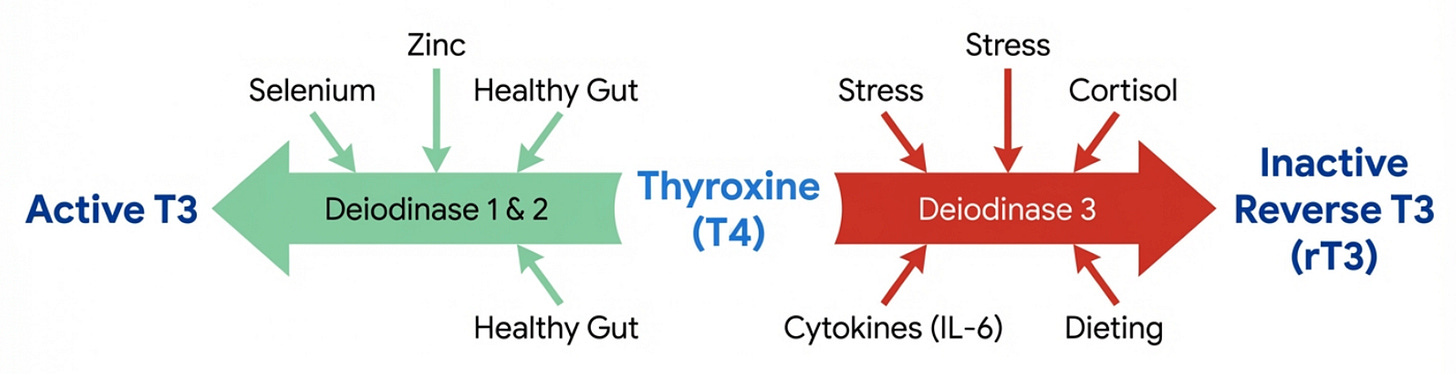
Thyroxine, T4, is a storage hormone. It does nothing until it is converted. The enzyme iodothyronine deiodinase cleaves one iodine atom from T4 to produce triiodothyronine, T3, the active signaling molecule that enters the cell nucleus, binds to thyroid hormone receptors, and initiates the transcription of genes governing metabolism, cognition, mood, and cardiovascular function. When conventional medicine prescribes levothyroxine monotherapy, it assumes that this conversion pathway is universally intact.
It is not. The conversion pathway is exquisitely fragile, and modern life attacks it from multiple directions simultaneously. It fails:
Under chronic stress, as elevated cortisol directly suppresses the T4-to-T3 conversion enzymes
Under caloric restriction, which downregulates deiodinase activity as a famine-adaptation signal
In the presence of gut dysbiosis, which impairs the intestinal conversion of T4 to T3 and elevates the inflammatory cytokines that suppress deiodinase function
In states of selenium deficiency, which disables the selenoprotein enzymes at the catalytic core of the entire pathway
Under elevated allostatic load, which runs all the above mechanisms concurrently
Selenium is not optional here. The deiodinase enzymes are selenoproteins: selenium sits directly at their catalytic core. In selenium deficiency, type-1 deiodinase activity in the liver and kidneys plummets, circulating T4 rises, and active T3 falls. A 2024 meta-analysis confirmed that selenium supplementation significantly reduces thyroid peroxidase antibody concentrations in patients with Hashimoto’s, but its role in conversion efficiency is arguably more fundamental than its role in autoimmunity. Without adequate selenium, the patient’s own thyroid hormone becomes essentially unusable.
Zinc, iron, and vitamin D complete the cofactor picture. Ferritin below 70 ng/mL, a level many physicians consider acceptable, correlates with impaired conversion and persistent hypothyroid symptoms even in patients on levothyroxine. The body’s ability to make and use thyroid hormone is inseparable from nutritional status, and nutritional status is inseparable from the conditions of modern life: ultra-processed food, depleted soil, and chronic inflammation.
Then there is reverse T3. Under conditions of chronic stress, prolonged caloric restriction, or high allostatic load, the body preferentially converts T4 not into active T3, but into reverse T3, a molecule that occupies the same receptor sites without activating them. It is a physiological brake, an ancient survival mechanism designed for famine and acute threat. Chronically stressed, chronically under-slept, chronically under-nourished people in the modern world are running on this brake continuously. Their TSH looks fine. Their tissues are in hibernation.
Layered beneath all of this is a genetic dimension that conventional endocrinology has been almost entirely unwilling to engage. The DIO1 and DIO2 genes encode the type-1 and type-2 deiodinase enzymes, and common polymorphisms in both have measurable consequences for thyroid hormone availability at the tissue level. The DIO2 Thr92Ala variant, present in roughly 16 percent of the population in homozygous form, reduces type-2 deiodinase activity in the pituitary and peripheral tissues, meaning carriers convert T4 to T3 less efficiently even under optimal nutritional conditions. A 2017 study in the Journal of Clinical Endocrinology and Metabolism found that thyroidectomized patients carrying this variant had significantly lower serum T3 levels despite being on standard levothyroxine replacement, and reported worse psychological well-being than non-carriers on identical doses. Their TSH was normalized. Their biology was not. DIO1 variants, meanwhile, influence the T3/T4 ratio in circulation, with certain polymorphisms consistently associated with lower free T3 relative to T4, a pattern that persists regardless of TSH. Taken together, these variants offer a genetic explanation for why a subset of patients on levothyroxine continue to feel profoundly unwell by every subjective measure while their labs are called perfect.
Return for a moment to the woman in my office. She is not struggling because she lacks willpower or resilience. She sleeps nine hours and wakes unrefreshed because her brain is operating in a state of hormonal deprivation that no standard lab panel has been designed to detect. The word retrieval failures, the slowing, the flat affect that her primary care physician interpreted as depression: these are not psychiatric symptoms. They are neurological ones, and their origin is upstream of the prescription pad.
This becomes particularly consequential when we consider what the research now calls isolated brain hypothyroidism. The brain is not simply a downstream recipient of circulating thyroid hormone. It is a semi-autonomous hormonal environment, separated from systemic circulation by the blood-brain barrier and entirely dependent on two specialized membrane transporters, MCT8 and OATP1C1, to shuttle T4 and T3 into neural and glial tissue. The brain relies primarily on type-2 deiodinase for local T4-to-T3 conversion, the same enzyme compromised in DIO2 Thr92Ala carriers. When peripheral conversion is poor, when neuroinflammation downregulates MCT8 or OATP1C1, or when a patient’s deiodinase genetics are working against them, the central nervous system can be functionally hypothyroid while the pituitary, operating with its own privileged enzymatic supply, signals that everything is fine. The cognitive symptoms, the word retrieval failures, the processing slowing, the affective flattening, the depression that does not lift with antidepressants: these are not vague or psychosomatic. They are the signature of a brain that is not receiving the T3 it requires to maintain myelination, synaptic plasticity, and neurotransmitter synthesis. The serum TSH, by design, cannot see any of this.
The body is not malfunctioning. It is responding rationally to irrational conditions. The system has not failed. It has adapted.
This is where the allostatic load framework, which I have written about extensively in this series, becomes clinically indispensable. High allostatic load drives elevated cortisol levels, which directly suppress T4-to-T3 conversion and promote the reverse T3 shunt. Stress physiology and thyroid physiology are not separate problems. They are the same problem viewed through different lenses.
The Autoimmune Blind Spot
Hashimoto’s thyroiditis is the most common thyroid condition in the developed world. It is also the most missed, diagnosed years or even decades after the immune attack on the gland has begun. The reason is simple: the standard diagnostic algorithm waits for TSH to break the threshold before ordering the antibody tests that would have identified the disease years earlier.
Thyroid peroxidase antibodies (TPOAb) and thyroglobulin antibodies (TgAb) are measurable long before the gland fails. They represent an early warning system, the immune system announcing its intentions. Most physicians do not order them until the TSH already signals overt disease. By then, the damage is done.
The more important reframe is this: Hashimoto’s is not primarily a thyroid disease. It is an autoimmune disease that targets the thyroid. Treating the thyroid without addressing the immune dysregulation that is destroying it is like mopping the floor while the pipe is still leaking. The pipe is the immune system. The inflammation driving antibody production has upstream causes worth understanding and intervening in, including gut permeability, gluten sensitivity, and chronic low-grade systemic inflammation.
The gluten-thyroid relationship warrants honest discussion. Gliadin, the primary immunogenic protein in wheat, shares structural similarity with thyroid peroxidase, a phenomenon known as molecular mimicry. When a genetically susceptible gut becomes permeable, gliadin antigens enter systemic circulation and may trigger antibody cross-reactivity against thyroid tissue. Multiple studies in non-celiac Hashimoto’s patients have reported reductions in TPOAb following a gluten-free diet, though the evidence remains heterogeneous and effect sizes vary. I present this not as settled science but as a clinical signal worth taking seriously in symptomatic patients with elevated antibodies and no other explanation.
Plasma zonulin, a biomarker of intestinal permeability, is significantly elevated in patients with Hashimoto’s compared with controls. Small intestinal bacterial overgrowth is associated with a 2.53-fold higher risk of developing Hashimoto’s. The gut-thyroid axis is mechanistic, bidirectional, and poorly appreciated in conventional endocrinology.
The Symptoms That Do Not Make the Differential
Word retrieval slows. Processing speed drops. Working memory becomes effortful in ways it never was before. These are thyroid-sensitive neurological functions, documented in subclinical hypothyroidism across multiple studies, and they are almost never attributed to the thyroid when a woman presents with them. She is told she is tired, stressed, perimenopausal, or anxious. She is rarely told that her T3 levels may be suboptimal.
The thyroid-depression overlap is particularly costly. T3 directly modulates serotonergic signaling in the prefrontal cortex and limbic system. Hypothyroid patients show reduced serotonin synthesis, altered receptor sensitivity, and blunted response to SSRIs. The STAR*D trial, the largest antidepressant effectiveness study ever conducted, found that T3 augmentation outperformed lithium as an add-on strategy in treatment-resistant depression. Adjunctive liothyronine significantly enhances treatment response in patients who have failed two prior antidepressant trials. These findings have not meaningfully changed the diagnostic algorithm. Women with thyroid-driven depression continue to receive antidepressant monotherapy while the underlying hormonal driver goes unaddressed.
Metabolically, thyroid insufficiency does not merely slow weight loss. It renders conventional weight-loss advice functionally useless for some patients. Baseline free T3 levels predict weight-loss outcomes. Patients on levothyroxine show attenuated response to dietary intervention compared to euthyroid controls, even when their TSH is normalized. Telling a woman with cellular hypothyroidism to eat less and move more is not wrong. It is simply incomplete in a way that feels like being told the problem is willpower.
Subclinical hypothyroidism is associated with diastolic dysfunction, elevated homocysteine, and dyslipidemia. The cardiovascular consequences of underdiagnosed thyroid disease accumulate quietly, over years, while the TSH remains “normal.”
What Functional Optimization Looks Like
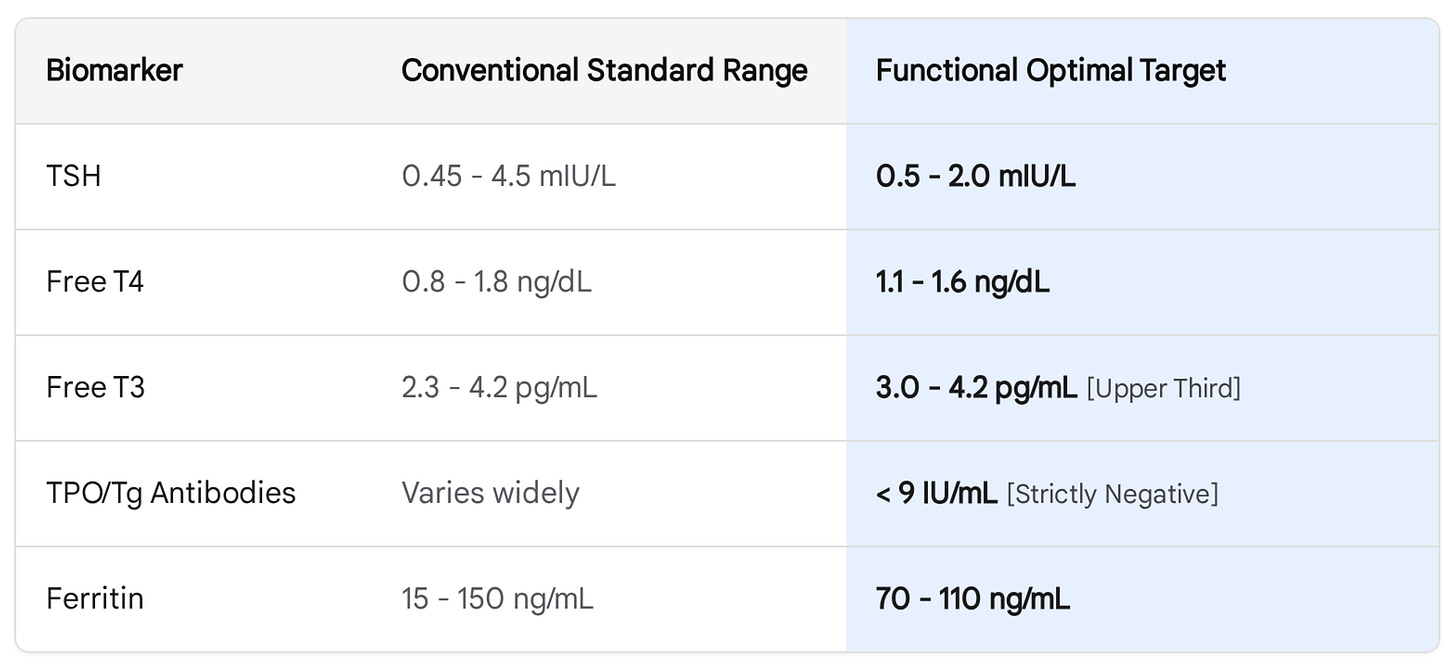
A complete thyroid evaluation includes TSH, free T4, free T3, reverse T3, thyroid peroxidase antibodies, and thyroglobulin antibodies. This is not a boutique panel. It is the minimum information required to understand what the thyroid system is doing at the cellular level.
In symptomatic patients, the functional target for TSH is 1.0 to 2.0 mIU/L, not “anywhere under 4.5.” Free T3 should sit in the upper third of the reference range. A normal TSH with a low-normal free T3 is not a clean bill of health. It is a conversion problem.
Nutritional optimization is not adjunctive. It is foundational. Selenium at 200 mcg daily reduces TPO antibody concentrations and supports deiodinase function. Zinc, often depleted in chronically stressed or gut-compromised patients, is required for T3 receptor binding. Ferritin should be optimized above 70 ng/mL, not merely above the anemia threshold. Vitamin D deficiency correlates with the prevalence of autoimmune thyroid disease and should be corrected before any pharmaceutical intervention is considered.
Gut rehabilitation belongs in the thyroid protocol. Addressing dysbiosis, reducing intestinal permeability, and evaluating for SIBO are not peripheral concerns. Given the bidirectional relationship between microbiome health and thyroid function, the gut is often where the most durable clinical gains are made.
When T3 supplementation is appropriate, either as liothyronine added to levothyroxine or as natural desiccated thyroid, the conversation most endocrinologists will not initiate is worth having. A randomized crossover trial published in the Journal of Clinical Endocrinology and Metabolism found that a significant subset of hypothyroid patients preferred desiccated thyroid extract over levothyroxine and lost more weight on it. The DIO2 Thr92Ala polymorphism, present in a meaningful portion of the population, specifically impairs type-2 deiodinase activity, meaning these patients cannot reliably convert T4 to T3 in the pituitary or peripherally at normal efficiency. They need T3 directly. TSH-based dosing will never identify them.
The thyroid does not gaslight. The framework does.
This is not an indictment of individual physicians. The physicians who looked at that TSH of 2.8 and said “you’re fine” were working within the system they were trained in, a system optimized for population-level efficiency, not individual physiological complexity. TSH threshold medicine handles millions of patients with minimal error at the population level. It handles the individual patient in my office with consistent, measurable inadequacy.
She is not a population. She is a system. And her system has been speaking clearly for three years. The problem was never that the signal was absent. The problem was that the instrument we were using was not designed to hear it.
We have better instruments now. The question is whether we are willing to use them.
Health is a verb.




The trick is how to find a doctor to help.
What a fantastic read. It gave me confidence to address this with my doctor. At 50 had a complete removal of thyroid for a small tumor. Now I fight every time I go in for a med renewal. I ask for 2 prescriptions of Synthroid having researched the seasonality of hormones in general. In winter I need more thyroid meds due to cold and dark and change in nutrition. In Summer I switch to a lower dose as the day’s lengthen and diet changes.
This has worked for me but every new doctor that I see fights me on that concept. Like I don’t know what optimizes my health.
Thank you for this great information.